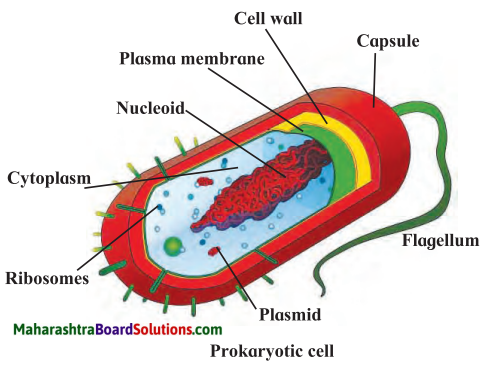
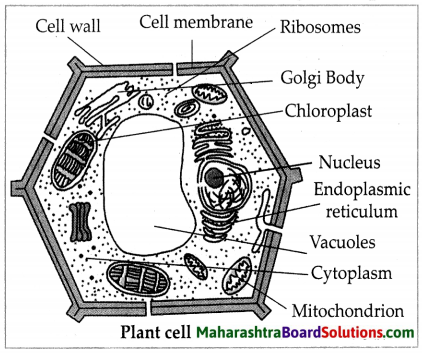
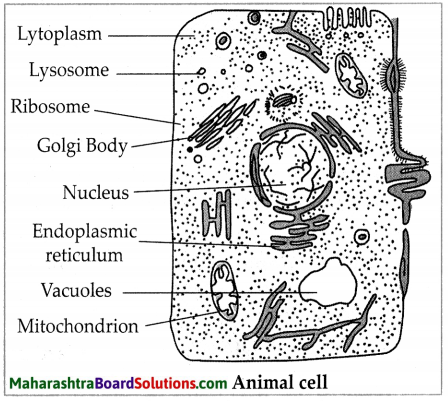
Balbharti Maharashtra State Board Class 7 Science Solutions Chapter 16 Natural Resources Notes, Textbook Exercise Important Questions and Answers.
Maharashtra State Board Class 7 Science Solutions Chapter 16 Natural Resources
Class 7 Science Chapter 16 Natural Resources Textbook Questions and Answers
1. Describe natural resources with reference to the following 3 types:
Question a.
Mineral resources
Answer:
Mineral wealth has an important place among natural resources. The rocks on the earth are mainly made of minerals. These minerals can be obtained by mining. Minerals that contain a high proportion of metals are ores, (i) Minerals are formed from the magma in the earth’s crust and the lava from the eruption of volcanoes when they cool and get transformed into crystals e.g. magnetite, mica, (ii) Minerals get transformed from one form into another due to large changes in temperature and pressure e.g. Diamond, Graphite, (iii) There are 3 types of minerals according to their properties Non-metal minerals: e.g. mica, sulphur, potash, diamond.
- Metals minerals: e.g. Iron, gold, silver, tin, bauxite, platinum.
- Energy minerals: e.g. Coal, mineral oil, natural gas, diamond, ruby, sapphire, jade are used as gems.
Some important minerals and ores are Iron ore, manganese, bauxite, copper and mica.
![]()
Question b.
Forest resources
Answer:
- A forest is a natural habitat of plants, animals and microbes.
- Forest perform certain specific protective and productive functions.
- We get different types of wood and many medicinal plants from forests.
- Forest wealth includes fire wood, trees like teak, mahogany, neem, acacia, subabhul, fibers, paper, rubber, gum, aromatic substances.
- We get fragrant essential oils from lemon grass, vanilla, kewada, vetiver (khus) and eucalyptus.
- Sandalwood and oil of eucalyptus are used for making soaps, cosmetics and incense sticks.
- Also we get fruits, bulbs, roots, honey, sealing wax, dyes from forest.
- Many medicinal plants adulsa, bel, neem, periwinkle, cinnamon, are used for treatment of various diseases such as cough cold, malaria, diarrhoea, cancer, fever and cold.
- Wood is used as a source of energy for cooking purpose and for keeping warm.
- For making furniture, tool handles, matches, bridges, boats etc.
- Bamboos are used for matting, flooring, basket, ropes, rafts, cots etc.
- Bamboos, wood are used in the manufacture of rayon, yams, artificial silk-fibers.
Question c.
Ocean resources
Answer:
- Oceans occupy about 70% of a earth’s surface.
- Energy can be obtained on a large scale from oceans.
- Sea waves at high and low tide and ocean currents are being used for generation of power.
- There are billions of tons of minerals dissolved in ocean water.
- There are large reserves of tin, chromium, potassium, phosphates, copper, zinc, iron, lead, thorium, manganese, sulphur, uranium in the ocean and sea beds.
- Also we get many types of gems, conches, shells and pearls from the sea.
- There are large reserves of mineral oil and natural gas at the bottom of sea.
- We get fishes, dried shrimp, shells, fungi, shark, cod fish and sea cucumber from the ocean.
![]()
2. Write answers to the following questions in your own words.
Question a.
What is meant by fossil fuel? What are their types?
Answer:
- Fossil fuels (such as coal or natural gas) are formed in the earth from dead plants and animals, due to high pressure from above and the heat from the earth’s interior.
- The dead plants, animals get transformed into fossil fuel.
- There are three types of fossil fuels which can be used for energy production, Coal, oil and natural gas.
a. Coal:
- Coal is a solid fossil fuel formed over millions of years by the decay of land vegetation,
- Coal is a store house of carbon. Coal is found in mines,
- It is like porous, hard, black rock.
- It is burnt to obtain heat energy.
- It is used as a fuel in thermal power plants and to run boilers and railway engines, for cooking and baking bricks.
- Anthracite is the coal of the highest grade.
- Producer gas and water gas are obtained from coal.
b. Mineral oil:
- Mineral oil is the liquid fuel formed by the decomposition of organic substances buried underground,
- The underground mineral oil is extracted through oil wells.
- Mineral oil is found mainly in oil sands, shale, sandstone and limestone at a depth of about 1000 to 3000 metres.
- Mineral oil is also known as petroleum or crude oil. It is greenish-brown in colour.
- Petroleum is a mixture of many compounds mainly of the hydrocarbon type.
- It also contains compounds of oxygen, nitrogen and sulphur.
- Petroleum is extracted through oil wells and refined by fractional distillation to separate other components,
- Aviation petrol, gasoline, diesel, kerosene, naphtha, lubricating oil, tar are all obtained from petroleum.
- They are used as fuel and for production of dyes, pesticides, perfumes and artificial fibres.
c. Natural gas:
- Natural gas is an important fossil fuel.
- It is lighter than air. It is found with petroleum in underground oil wells and in some places as natural gas alone.
- The main component of natural gas is methane (CH4), ethane (C2H6), propane (C3H8) and butane (C4H10) are present in small proportions.
- This gas is highly flammable.
- This fuel can be carried over long distance by means of a gas pipeline.
- In absence of pipeline, it is transformed under high pressure into compressed natural gas (CNG) and liquefied natural gas (LNG).
![]()
Question b.
Make a list of the components we obtain from mineral oil.
Answer:
- Mineral oil is the liquid fuel. It is also known as petroleum or crude oil. Petroleum is refined by fractional distillation to separate other components.
- Aviation petrol, gasoline, diesel, kerosene, naphtha, lubricating oil, tar are the components we obtain from mineral oil.
Question c.
What do we get from forest?
Answer:
- A forest is a natural habitat of plants, animals and microbes. We get wood, firewood from trees like teak, mahogany, neem, subabhul.
- Also we get fibers, paper, rubber, gum and aromatic substances.
- We get many medicinal plants like Adulsa, Bel, Neem, Periwinkle, Cinnamon, Cinchona which are used for treatment of various diseases like cough, cold, diarrhea, fever, cancer, diarrhoea, nausea, malaria.
- We get fragrant essential oils from lemon grass, vanilla, kewada, khus, eucalyptus.
- In addition to this we get fruits, bulbs and roots, honey, sealing wax, catechu, dyes from forest.
![]()
Question d.
What are the items included in ocean resources? What are their uses?
Answer:
- Tons of minerals are found in ocean water.
- They are tin, chromium, phosphates, copper, zinc, iron, lead, manganese, sulphur, uranium etc.
- We also get many types of gems, conches, shells and pearls from the sea.
- Mineral oil and natural gas is found at the bottom of sea.
Bio-resources in oceans
- Fishes – like pomfret, seer fish as also shrimps
- prawns – they are sources of proteins and vitamins, therefore, mainly used as sea food
- Dried shrimp, Bombay duck powder – used as poultry feed and is a good manure.
- Shells – used for preparation of medicines, ornaments and decorative articles.
- Fungi – used for the production of antibiotics.
- shark and cod fish – used for producing edible oil rich in Vitamins A, D and E.
- Sea cucumbers – used as medicine for treating cancer and tumours.
Mineral resources from oceans
- Thorium – used in the production of atomic energy.
- Magnesium – used in the flash bulb of a camera.
- Potassium – the main ingredient in production of soap, glass, fertilizer.
- Sodium – used in the production of cloth and paper.
- Sulphate – used in making artificial silk.
![]()
Question e.
Why should we prevent the wastage of fuels used for vehicles?
Answer:
- Vehicles are powered by gasoline or diesel petrol.
- Nowadays compressed natural gas (CNG) is also used.
- Aviation petrol, gasoline, diesel, we get from petroleum after fractional distillation.
- Petroleum is a non-renewable source of energy.
- It is getting depleted day by day and the demand for fuel has increased greatly due to the rapidly growing population.
- But the reserves of petroleum (fossil fuel) are limited.
- It is becoming difficult to meet the increased demand. So we should prevent the wastage of fuel.
Question f.
Why is the diversity of plants and animals in the forests declining?
Answer:
- The major causes of biodiversity decline are land use changes, pollution, changes in atmosphere, CO2 concentration, changes in the nitrogen cycle, acid rain, climate alteration and introduction of exotic species.
- All this is due to population growth.
- This is the main cause of the loss of biodiversity. More of wood for fuel is been used, more land is required for food production, livestock grazing etc.
- Rainforest land is converted to industrial and residential usage, so natural habitat of birds, animals is destroyed.
- Due to population growth most of wild land is converted to crop land. So no space for animals to live, their natural habitat is destroyed.
![]()
Question g.
Write the names of five minerals and the useful substances obtained from them.
Answer:
Some important minerals and ores are:
- Iron ore: It is used for making farming implements, rails of railway tracks etc.
- Manganese: Compounds of manganese are used in the preparation of medicines and for giving a pink tinge to glass. Manganese is also used in electrical appliances.
- Bauxite: It contains 55% Aluminium. Therefore, it is used mainly in aeroplanes, transport vehicles and to make electric wire.
- Copper: It is used to make electric wires as well as in radios, telephones, vehicles, and for making kitchen utensils and statues.
- Mica: Mica has many uses such as in ayurvedic medicines, dyes, electric machines and equipment, wireless communication equipment, etc.
![]()
Question h.
Name the two important stages in the process of obtaining metals from ores.
Answer:
The two important stages in the process of obtaining metals from ores are extraction and purification.
3. What steps are taken for protection and conservation of natural resources?
Question a
What steps are taken for protection and conservation of natural resources?
Answer:
- Protection and conservation of natural resources is necessary to protect the natural world. (CNRM) Community Natural Resource Management has been promoted in recent years for biological conservation.
- The World Conservation Union has been formed. It is the world’s leading authority and democratic platform in conservation and sustainability.
- Young trees should not be cut. More and more trees need to be grown.
- The stringent restrictions/ laws/ Regulations regarding use of forests should be strictly followed. Awareness is being created through Mass media.
![]()
4. Complete the Flow Chart:
Question a.
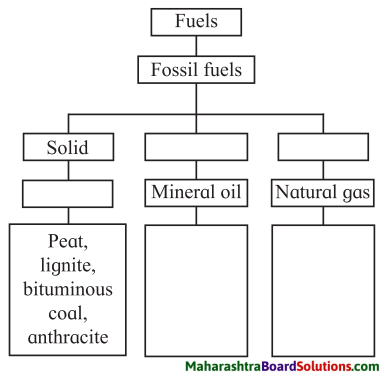
Answer:
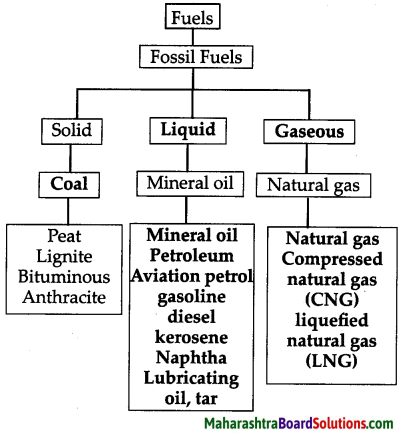
5. How does the economic condition of a nation depend on its natural resources?
Question a.
How does the economic condition of a nation depend on its natural resources?
Answer:
1. Natural resources are a necessary condition for economic growth.
2. Three circles enclosed within one another shows how both economy and society are subsets of our planetary ecological system.

3. Natural resource economics deals with the supply, demand and allocation of the Earth’s natural resources.
4. Economic growth is an increase in the value of goods and services produced in an economy.
5. The natural resources of a country depend on the climatic and environmental conditions.
6. Countries having plenty of natural resources enjoy good growth than countries with small amount of natural resources.
7. A country having skilled and educated work force with rich natural resources takes the economy on the growth path, because skilled and educated people can efficiently utilize or exploit natural resources, e.g. Saudi Arabia: their economic growth is high, because they have oil wells.
8. All countries import fuel or oil from them so Saudi Arabia is economically a rich country.
6. Which medicinal plants will you grow on your school premises and near your house? Why?
Question a.
Which medicinal plants will you grow on your school premises and near your house? Why?
Answer:
1. I will grow tulsi, neem, lemon grass, bel, adulsa, periwinkle, cinnamon, ashwagandha, shatavari, amla, hirda, behda.
2. These plants are used for treatment of various diseases.
- Tulsi → for cough and cold.
- Amla, hirda, behda → Help in digestion of food or any stomach related problems.
- Adulsa → for cough and cold
- Bel → for Diarrhoea
- Neem → Fever and cold
- Periwinkle → Cancer
- Cinnamon → Diarrhoea, nausea.
- Cinchona → Malaria.
- Ashwagandha → It can reduce blood sugar levels. It can reduce stress and anxiety, may reduce symptom of depression.
- Shatavari → has been used for centuries in Ayurveda to support the reproductive system.
- Amla → Excellent source of vitamin C. Amla juice is best tonic to keep you younger for long.
- Hirda → This fruit is commonly called Haritaki. It is used for treating arthritis, dental problems like caries, bleeding gums.
- Behda →(Beleric) is a rejuvenative and laxative, proves beneficial for hair, throat and eyes.
- Tulsi → Tulsi has germicidal, fungicidal, anti-bacterial anti-biotic properties, cures fever. Treats diabetes, protects the heart. Tulsi has anti-oxidant component Eugenol which keeps one’s blood pressure under control.
![]()
Project:
Question 1.
Collect conches and shells of various shapes and colours and make a decorative article.
Question 2.
Collect information about the mines of various minerals.
Class 7 Science Chapter 16 Natural Resources Important Questions and Answers
Fill in the blanks:
Question 1.
………….. is the major raw material for biogas.
Answer:
cow dung
Question 2.
Atomic energy is obtained by using ores of ………….. .
Answer:
Uranium
![]()
Question 3.
Biogas generation is mainly based on the principle of ………….. .
Answer:
Fermentation
Question 4.
Floods can be prevented by ………….. .
Answer:
Afforestation
Question 5.
Coal, petroleum, natural gas are ………….. resources.
Answer:
non-renewable
Question 6.
The total percent of land of world under forest cover is ………….. .
Answer:
30%
Question 7.
A naturally occurring substance ………….. .
Answer:
minerals
Question 8.
Minerals that contain a high proportion of metal are called ………….. .
Answer:
ore
![]()
Question 9.
Coal, mineral oil and natural gas are ………….. .
Answer:
fossil fuel
Question 10.
Deposits of common salt are also found in the earth. This salt is called ………….. .
Answer:
rock salt
Question 11.
………….. is the liquid fuel formed by the decomposition of organic substances.
Answer:
Mineral oil
Question 12.
The underground mineral oil is extracted through ………….. .
Answer:
oil wells
![]()
Question 13.
Mineral oil is also known as ………….. or ………….. .
Answer:
petroleum, crude oil
Question 14.
………….. are the preserved remains of dead organisms in rock.
Answer:
Fossil
Question 15.
Coal can be formed from ………….. .
Answer:
Fossils
Question 16.
Impurities of sand and soil in ore are called ………….. .
Answer:
Gangue
Question 17.
Coal mainly contains ………….. .
Answer:
carbon
![]()
Question 18.
Petroleum is formed from ………….. .
Answer:
organisms in sea
Question 19.
Separation of various fractions of petroleum is called ………….. .
Answer:
Refining
Question 20.
We can obtain minerals from rocks by ………….. .
Answer:
mining
Question 21.
………….. is the most important ore of aluminium
Answer:
Bauxite
Question 22.
………….. is the coal of the highest grade.
Answer:
Anthracite
![]()
Question 23.
Metals are obtained from their ore by ………….. and ………….. .
Answer:
extraction, purification
Question 24.
………….. is the largest oil and gas research and production company in India.
Answer:
ONGC
Question 25.
Natural gas is transformed under high pressure into ………….. and ………….. .
Answer:
LNG and CNG
![]()
Question 26.
The main component of Natural gas is ………….. .
Answer:
Methane
Question 27.
………….. and ………….. are used for adding fragrance in soaps and incense sticks.
Answer:
Sandalwood and oil of Encalyptus
Question 28.
………….. is a natural herbal medicine for cough and cold.
Answer:
Adulsa
![]()
Answer the following:
Question 1.
What is meant by natural resources?
Answer:
- We get many substances from nature.
- They satisfy a variety of our daily needs. Soil, stones, minerals, air, water, plants and animals on the earth are all various kinds of natural resources.
Question 2.
What are alternative fuels?
Answer:
Hydrogen, biofuels, methanol or wood alcohol, ethanol or green alcohol are some of the alternative fuels.
![]()
Question 3.
What is meant by fuels?
Answer:
- Fuel is a substance that is used in day-to-day life and burned to generate energy.
- Fuels such as coal, wood, oil, gas provide energy when burned.
- Fuels are found in solid, liquid or gaseous state.
Question 4.
Which natural resources do we use as fuels?
Answer:
We use coal, mineral oil and natural gas as fuels.
Question 5.
What is meant by forests?
Answer:
An extensive area of land covered by a variety of plants is called a forest. A forest is a natural habitat of plants, animals and microbes.
![]()
Question 6.
What are the uses of forests?
OR
Write short note on forest and its uses.
Answer:
- A forest is a natural habitat of plants, animals and microbes.
- Forest help improve and maintain the quality of environment.
- Forest helps in prevention of soil-erosion, help to control floods, protects wild life, maintains the balance of atmospheric gases.
- We get many medicinal plants from forest which help in treatment of various diseases.
- Examples Adulsa, Bel, Neem, periwinkle, amla tulsi, cinnamon.
- We also get wood, firewood from trees like teak, mahogani, neem, sababhul. Wood is useful for making furniture, farming implements, in construction work.
- Forest wealth includes fibres, paper, rubber, gum and aromatic substances.
- We get fragrant essential oils from lemon grass, vanilla, kewada and eucalyptus.
- They are used for making soaps, cosmetics and incense sticks. We get various fruits, bulbs, roots, honey, sealing wax, catechu dyes etc. from forest.
Question 7.
Are minerals to be found in seas and on the seabed as they are found inside the earth?
Answer:
Yes, there are very large reserves of tin chromium, phosphates, copper, zinc, iron, lead, manganese, sulphur, uranium etc. in the ocean and seabed.
![]()
Question 8.
How is mineral oil formed?
Answer:
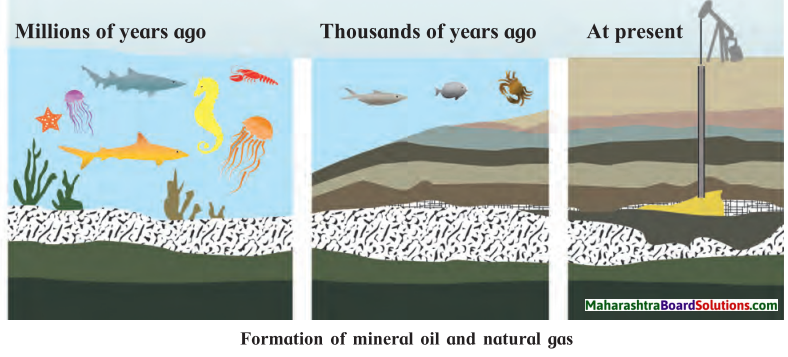
- Mineral oil is the liquid fuel formed by the decomposition of organic substances buried underground.
- Millions of years ago, bodies of dead sea organisms sank to the bottom of the sea.
- Layers of soil and sand collected on them.
- Due to high pressure and temperature the remains of the dead organisms were transformed into mineral oil.
- The underground mineral oil is extracted through oil wells.
![]()
Use your brain power!
Question 1.
Why are all minerals not ores?
Answer:
- Minerals that contain a high proportion of metal are called ores.
- Majority of the metals occur in the form of compounds called ores.
- But all minerals are not ores.
- Few metals like gold, silver, copper, platinum and bismuth occur in free state in nature. So all minerals are not ores.
Question 2.
What is meant by metal mineral and non¬metal mineral?
Answer:
- Metal-minerals contain metallic elements in their chemical formula. So they are called metal-minerals, for example, iron, copper, gold, silver etc.
- They contain metal in raw form.
- Non-metallic minerals do not contain metal elements in their inorganic chemical formula Example, Clay, Diamond, Dolomite, Gypsum, Mica, Quartz.
![]()
Question 3.
Why is mineral oil called ‘liquid gold’?
Answer:
Because of its high price and value and its economic need, the mineral oil is called liquid gold. It’s reserve is less and demand is more.
Question 4.
Why is coal called black gold?
Answer:
- Coal is a storehouse of carbon. Coal is used as a fuel, in thermal power plants as well as to run boilers and railway engines.
- It is used as a fuel for cooking and for baking bricks in kilns on a large scale.
- The gaseous fuels, producer gas and water gas are obtained from coal.
- Coal as an energy resource, contributes greatly to industrial development.
- As coal is black in colour and worth a lot of money just like gold, it is called black gold.
Question 5.
What would happen if underground mineral resources are exhausted?
Answer:
- Coal, petroleum and other mineral are all non-renewable sources of energy.
- The reserve is less and demand is more. They would get exhausted one day.
- If such a situation occurs, we will not get electricity.
- All our household and industries depend on electricity.
- Transportation and household items, and food items would become very costly.
- The survival of human life will be ver\ difficult.
![]()
Question 6.
Why is natural gas an eco-friendly fuel?
Answer:
- Natural gas is an important source of energy which is eco-friendly because of its low carbon dioxide emission.
- When burned it produces 45% less CO2 than coal, 30% less than oil, and about 15% less than wood.
- It does not produce carbon monoxide, sulphur dioxide or airborne particles, all of which are harmful to human and animal health.
- It is referred as the clean energy source and is fuel for the present century as it does not ‘ pollute the atmosphere or contribute to global warming.
Question 7.
What useful things will we have to do without if rubber is no longer available?
Answer:
- Rubber has played a largely hidden role in global environment history for more than 150 years.
- In industries, rubber is needed to connect and protect all moving parts of machines.
- Native people used rubber for rubber boots.
- Household uses rubber in everything viz dish washing gloves, toys, jar seals, tyres in vehicles.
- Welcome mat at the door, Boots, raincoats, mattress cushions, earplugs, hot water bottles, surgical tubing and gloves, in labs, birth control device.
- In schools rubber bands, erasers, mouse pads, key board, rolling chair wheels. Head phone pads, rubber stamps are useful items used in schools and offices.
![]()
Question 8.
What are the adverse effects of clearing of forest or cutting down trees?
Answer:
- Clearing of forest is also called Deforestation.
- Removal or cutting down of trees has resulted in damage to habitat, biodiversity loss and aridity.
- If has adverse impacts on concentration of atmospheric carbon dioxide. (Trees use up carbon dioxide from the atmosphere for the process of photosynthesis.)
- Deforestation is a main contributor to global warming. Major cause of enhanced greenhouse effect. Deforestation cause carbon dioxide to linger in the atmosphere.
- Deforestation reduced the content of water in the soil and ground water and atmospheric moisture, because trees extract ground water through the roots and release it into the atmosphere.
- When a part of forest is removed the trees no longer transpire this water, resulting in much drier climate.
- Due to deforestation soil erosion and flooding, landslide problems occur.
- Deforestation results in decline in biodiversity, on a natural global scale is known to cause the extinction of many species.
- Forest is habitat for wild life, and many medicinal plants. If forests are removed it will affect wild life and also we will not get medicinal plants.
Find out:
Question 1.
How did the various ages of the prehistoric period get their names on the basis of the uses of metals.
Answer:
1. The three-age system in history archaeology, and physical anthropology is a methodological concept adopted during the 19th century by which artifacts and events of late prehistory and early history could be ordered into a recognizable chronology.
2. Initially developed by C. J. Thomson, director of the Royal Museum of Nordic Antiquities, Copenhagen – as a means to classify the museum’s collection according to whether the artifacts were made of stone, bronze or iron.
3. Depending-upon the use of stone, bronze, iron, that period was known as stone age, bronze age, and iron age, respectively.
![]()
Answer the following:
Question 1.
Write down the difference between Metal minerals and Non-metallic minerals.
Answer:
| Metal minerals | Non-metallic minerals |
| 1. Metal minerals contain metal in raw form. | 1. Non-metallic minerals do not contain metal. |
| 2. These metals are generally associated with igenous rocks. | 2. These metals are generally associated with sedimentary rocks. |
| 3. They are usually hard and have shine of their own. | 3. They are not usually hard, have no shine of their own. |
| 4. e.g. Iron, copper tine, bauxite. | 4. e.g. Salt, coal, mica, clay. |
Question 2.
Write down the different types of coal.
Answer:
Peat, lignite (brown coal), bituminous coal and anthracite are various types of coal.
Anthracite is the coal of the highest grade.
![]()
Question 3.
Write down the characteristics of Compressed Natural Gas.
Answer:
Characteristics of CNG:
- Catches fire easily.
- No solid waste remains after combustion.
- Carbon dioxide and water are formed in small quantities.
- Other pollutants are not produced.
- Can be transported easily.
- Combustion can be controlled easily.
Observe the pictures and answer the question.

Question a.
What is the process shown in the given picture?
Answer:
The process shown is formation of mineral oil and natural gas.
![]()
Question b.
Explain the process.
Answer:
- Mineral oil is the liquid fuel formed by the decomposition of organic substances buried underground.
- Millions of years ago, bodies of dead sea organisms sank to the bottom of the sea.
- Layers of soil and sand collected on them.
- Due to high pressure and temperature the remains of the dead organisms were transformed into mineral oil.
- The underground mineral oil is extracted through oil wells.
Question c.
What are the gaseous substances formed in the above process?
Answer:
Natural gas is also formed along with mineral oil. It contains methane, ethane, propane and butane gases.
![]()
Question d.
When the products of the process is refined, what other components are produced?
Answer:
When mineral oil is refined by fractional distillation products produced are petrol, diesel, kerosene, naphtha lubricating oil, tar, etc.
 Answer:
Answer: